GENTLE/S:
Robotic assistance in neuro and motor rehabilitation
The Fifth Framework programme of the European Commission:
Quality of life and management of living resources - The ageing population and their disabilities
Cerebral vascular accidents such as ischaemic and haemorrhagic stroke are a leading cause of disability with a prevalence of about 140 per 10,000 of the population. The associated incidence is approximately 20 per
10,000 per year with two thirds of incidence in older people over 65 years of age. Of the survivors approximately one third recover to have mild impairments, another third are moderately impaired and the remainder
are severely impaired.
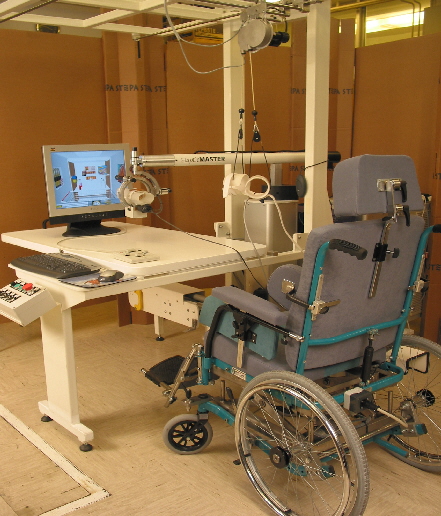
GENTLE/s has taken a rapid prototyping approach to developing the technology for machine mediated stroke rehabilitation. This allowed the technical partners to produce a viable clinical prototype within fourteen
months of the project start date and also allowed clinical partners to begin a pilot clinical study within the first half of the allotted project time. Results from this pilot study along with ergonomic information
on the use of the machine, was then available for analysis and to refine the design approach. After a revision of the design a more substantial clinical study was undertaken. A parallel activity was begun on the
kinematics and biomechanics of the person using the mechanism, which has allowed both a good understanding of the forces and range of movement involved. It proved possible to use robotic techniques to 'back
estimate' joint torques for a greater clinical understanding. An important aspect of the research was to assess not only the clinical impact but the readiness of the technology for a market that could be at a
hospital, a patient's home, a rural health-centre or a medical practice.
The project has identified the user needs via consultation with the user community and via a workshop with clinicians and people who have had a stroke to discuss the important aspects in the design of the
system. Two clinical prototypes produced and two clinical evaluation sites in two countries selected, the first at the Tallaghn Hospital, Dublin, and the second at the RBH-Battle Hospital in Reading. Partners from
these two organizations have developed a detailed experimental protocol and used this as the basis of a submission to the respective ethics committees.
Ethical approval has been granted for the study to proceed at one site, with the committees' decision pending for the second site.
Work was completed on a second phase prototype based on a modified HapticMaster haptic interface from Fokker Control. This prototype was fully validated both from a technical and a clinical aspect.
Dissemination activities included a poster, brochures and publications in both academic and general literature as well as two international television broadcasts on the project.
If you are interested in finding out more about the project and its partners, please contact
Dr William Harwin at the University of Reading's Department of Cybernetics.
|